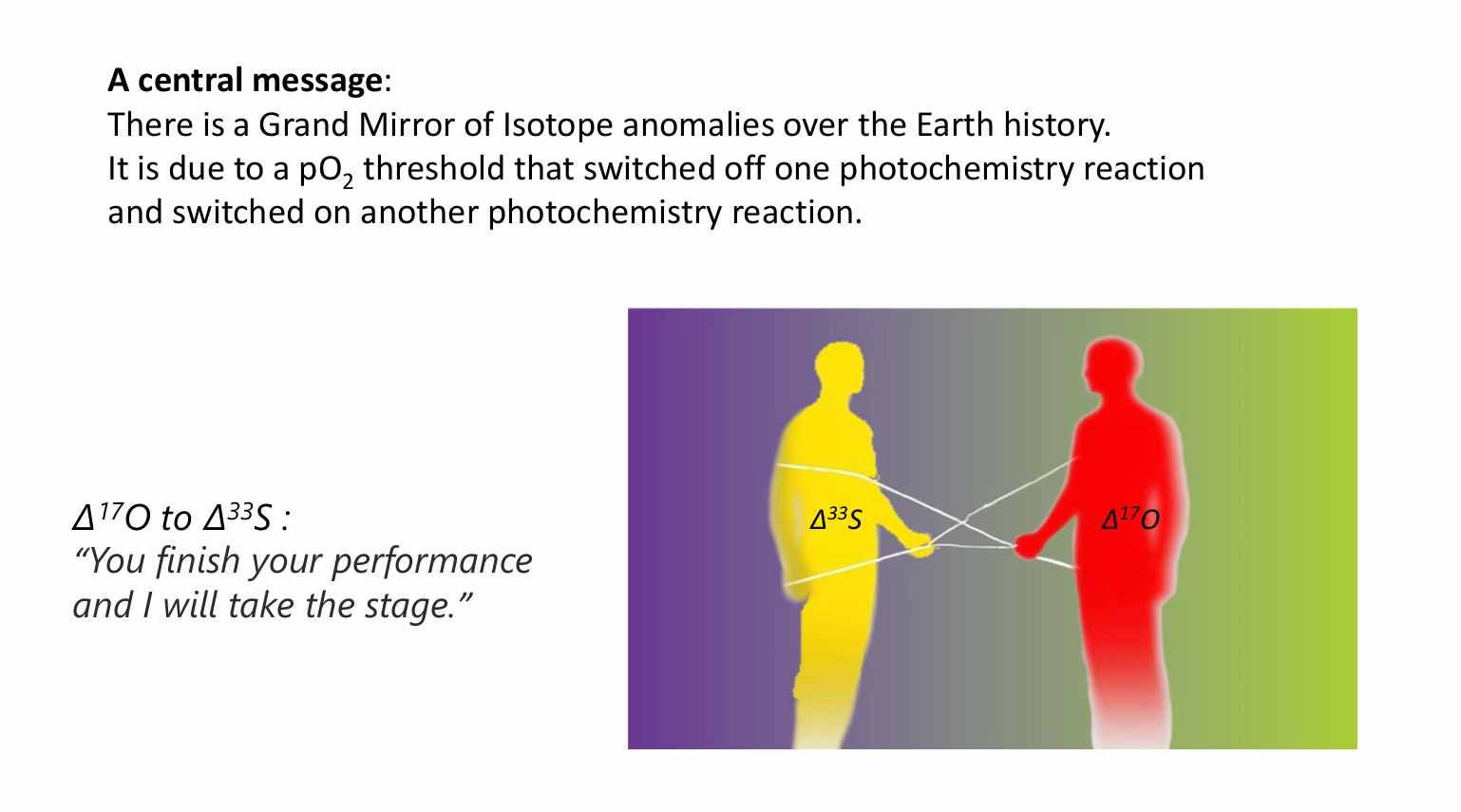
The existence of a pO₂ threshold that switched off one photochemical reaction, i.e., deep UV SO₂ photolysis in the troposphere, and switched on another photochemical reaction, i.e., the CO₂–O₂–O₃ reaction network in the stratosphere, has resulted in a grand mirror of isotope anomalies over the 4.6 Ga Earth history. The rock record reveals this grand pattern.
Financial support is provided by the National Natural Science Foundation of China (42494851, 42494852).
1. M. Domeier, V. Magni, M.W. Hounslow, and T.H. Torsvik, Episodic zircon age spectra mimic fluctuations in subduction, Sci. Rep. 8, 17471 (2018).
2. C.A. Fuchsman and E.E. Stüeken, Using modern low-oxygen marine ecosystems to understand the nitrogen cycle of the Paleo- and Mesoproterozoic oceans, Environ. Microbiol. 23, 2801 (2021).
3. Y.L. Yung, A.Y.T. Lee, F.W. Irion, W.B. DeMore, and J. Wen, Carbon dioxide in the atmosphere: isotopic exchange with ozone and its use as a tracer in the middle atmosphere, J. Geophys. Res. Atmos. 102, 10857 (1997).
4. H. Bao, J.R. Lyons, and C. Zhou, Triple oxygen isotope evidence for elevated CO₂ levels after a Neoproterozoic glaciation, Nature 453, 504 (2008).
5. H.D. Holland, The oxygenation of the atmosphere and oceans, Philos. Trans. R. Soc. B Biol. Sci. 361, 903 (2006).
6. D.T. Johnston, Multiple sulfur isotopes and the evolution of Earth’s surface sulfur cycle, Earth-Sci. Rev. 106, 161 (2011).
7. H. Bao, I.J. Fairchild, P.M. Wynn, and C. Spötl, Stretching the envelope of past surface environments: Neoproterozoic glacial lakes from Svalbard, Science 323, 119 (2009).
8. P.W. Crockford, J.A. Hayles, H. Bao, N.J. Planavsky, A. Bekker, P.W. Fralick, G.P. Halverson, T.H. Bui, Y. Peng, and B.A. Wing, Triple oxygen isotope evidence for limited mid-Proterozoic primary productivity, Nature 559, 613 (2018).
9. J.F. Busch, E.B. Hodgin, A.S.C. Ahm, J.M. Husson, F.A. Macdonald, K.D. Bergmann, J.A. Higgins, and J.V. Strauss, Global and local drivers of the Ediacaran Shuram carbon isotope excursion, Earth Planet. Sci. Lett. 579, 117368 (2022).
10. H. Wang, Y. Peng, C. Li, X. Cao, M. Cheng, and H. Bao, Sulfate triple-oxygen-isotope evidence confirming oceanic oxygenation 570 million years ago, Nat. Commun. 14, 4315 (2023).
11. G.A. Shields, B.J.W. Mills, M. Zhu, T.D. Raub, S.J. Daines, and T.M. Lenton, Unique Neoproterozoic carbon isotope excursions sustained by coupled evaporite dissolution and pyrite burial, Nat. Geosci. 12, 823 (2019).
12. H. Wang, C. Li, Y. Peng, J. Zhang, M. Cheng, X. Cao, W. Qie, Z. Zhang, M.S. Dodd, M. Hou, M. Wallace, A.V.S. Hood, T.W. Lyons, and H. Bao, Two-billion-year transitional oxygenation of the Earth’s surface, Nature 645, 665 (2025).
13. H. Bao, Sulfate: a time capsule for Earth’s O₂, O₃, and H₂O, Chem. Geol. 395, 108 (2015).
14. A.B. Romero and M.H. Thiemens, Mass-independent sulfur isotopic compositions in present-day sulfate aerosols, J. Geophys. Res. Atmos. 108, 4524 (2003).
15. J. Savarino, A. Romero, J. Cole-Dai, S. Bekki, and M.H. Thiemens, UV-induced mass-independent sulfur isotope fractionation in stratospheric volcanic sulfate, Geophys. Res. Lett. 30, 2131 (2003).
16. X. Han, Q. Guo, H. Strauss, C. Liu, J. Hu, Z. Guo, R. Wei, M. Peters, L. Tian, and J. Kong, Multiple sulfur isotope constraints on sources and formation processes of sulfate in Beijing PM₂.₅ aerosol, Environ. Sci. Technol. 51, 7794 (2017).
17. D. Hu, M. Li, X. Zhang, A.V. Turchyn, Y. Gong, and Y. Shen, Large mass-independent sulfur isotope anomalies link stratospheric volcanism to the Late Ordovician mass extinction, Nat. Commun. 11, 2297 (2020).
18. S.A. Young, K. Loukola-Ruskeeniemi, and L.M. Pratt, Reactions of hydrothermal solutions with organic matter in Paleoproterozoic black shales at Talvivaara, Finland: evidence from multiple sulfur isotopes, Earth Planet. Sci. Lett. 367, 1 (2013).
19. R. Li, S.-Z. Shen, X.-P. Xia, B. Xiao, Y. Feng, and H. Chen, Atmospheric ozone destruction and the end-Permian crisis: evidence from multiple sulfur isotopes, Chem. Geol. 647, 121936 (2024).
20. R.N. Mitchell, N. Zhang, J. Salminen, Y. Liu, C.J. Spencer, B. Steinberger, J.B. Murphy, and Z.-X. Li, The supercontinent cycle, Nat. Rev. Earth Environ. 2, 358 (2021).
21. R.D. Nance and J.B. Murphy, Origins of the supercontinent cycle, Geosci. Front. 4, 439 (2013).
22. M.R. Rampino, K. Caldeira, and S. Rodriguez, Sixteen mass extinctions of the past 541 My correlated with 15 pulses of Large Igneous Province (LIP) volcanism and the 4 largest extraterrestrial impacts, Glob. Planet. Change 234, 104369 (2024).
23.S.E. Grasby and D.P.G. Bond, How large igneous provinces have killed most life on Earth—numerous times, Elements 19, 276 (2023).
24. J. Farquhar, H. Bao, and M. Thiemens, Atmospheric influence of Earth's earliest sulfur cycle, Science 289, 756 (2000).
25. D.C. Catling and M.W. Claire, How Earth's atmosphere evolved to an oxic state: a status report, Earth Planet. Sci. Lett. 237, 1 (2005).
26. K.J. Zahnle, D.C. Catling, and M.W. Claire, The rise of oxygen and the hydrogen hourglass, Chem. Geol. 362, 26 (2013).
27. M.S. Duncan and R. Dasgupta, Rise of Earth’s atmospheric oxygen controlled by efficient subduction of organic carbon, Nat. Geosci. 10, 387 (2017).
28. D.A. Stolper, J.A. Higgins, and L.A. Derry, The role of the solid earth in regulating atmospheric O₂ levels, Am. J. Sci. 321, 1381 (2021).
29. M.M. Hirschmann, The deep Earth oxygen cycle: mass balance considerations on the origin and evolution of mantle and surface oxidative reservoirs, Earth Planet. Sci. Lett. 619, 118311 (2023).
30. N.F. Wogan, D.C. Catling, K.J. Zahnle, and M.W. Claire, Rapid timescale for an oxic transition during the Great Oxidation Event and the instability of low atmospheric O₂, Proc. Natl. Acad. Sci. U.S.A. 119, e2205618119 (2022).
31. A.A. Pavlov and J.F. Kasting, Mass-independent fractionation of sulfur isotopes in Archean sediments: strong evidence for an anoxic Archean atmosphere, Astrobiology 2, 27 (2002).
32. B. Luz, E. Barkan, M.L. Bender, M.H. Thiemens, and K.A. Boering, Triple-isotope composition of atmospheric oxygen as a tracer of biosphere productivity, Nature 400, 547 (1999).
33. E. Thomassot, J. O'Neil, D. Francis, P. Cartigny, and B.A. Wing, Atmospheric record in the Hadean Eon from multiple sulfur isotope measurements in Nuvvuagittuq Greenstone Belt (Nunavik, Quebec), Proc. Natl. Acad. Sci. U.S.A. 112, 707 (2015).
34. H. Bao, S. Yu, and D.Q. Tong, Massive volcanic SO₂ oxidation and sulphate aerosol deposition in Cenozoic North America, Nature 465, 909 (2010).
35. H. Bao and D.R. Marchant, Quantifying sulfate components and their variations in soils of the McMurdo Dry Valleys, Antarctica, J. Geophys. Res. 111, D16301 (2006).
36. T. Sun, H. Bao, M. Reich, and S.R. Hemming, More than ten million years of hyper-aridity recorded in the Atacama Gravels, Geochim. Cosmochim. Acta 227, 123 (2018).
37. I. Genot, D. Au Yang, E. Martin, P. Cartigny, E. Legendre, and M. De Rafelis, Oxygen and sulfur mass-independent isotopic signatures in black crusts: the complementary negative Δ³³S reservoir of sulfate aerosols?, Atmos. Chem. Phys. 20, 4255 (2020).
38. M. Baroni, M.H. Thiemens, R.J. Delmas, and J. Savarino, Mass-independent sulfur isotopic compositions in stratospheric volcanic eruptions, Science 315, 84 (2007).
The Grand Mirror of Isotope Anomalies-Supplementary Information
The Grand Mirror of Isotope Anomalies
Huiming Bao
1. International Center for Isotope Effects Research (ICIER), State Key Laboratory of Critical Earth Material Cycling and Mineral Deposits, Nanjing University, Nanjing 210023, China
2. Frontiers Science Center for Critical Earth Material Cycling, School of Earth Sciences and Engineering, Nanjing University, Nanjing 210023, China
There are often simple and broad patterns in a seemingly chaotic history of a complex system that is filled with random events. Earth history is not an exception. For example, driven by mantle convection and plate tectonics, Earth's continents have repeatedly assembled into supercontinents and then broken apart—the supercontinent cycle (~400–600 million years) [1-3]. Episodic large igneous provinces may have triggered several mass extinction events of macroscopic life forms in the Phanerozoic Eon (since 541 Ma) [4, 5]. Identifying general patterns like these not only helps to reveal deep, underlying mechanisms but also offers excellent pedagogical frameworks in exploring Earth history. Here I highlight another grand pattern: The Grand Mirror of Isotope Anomalies (GMIA).
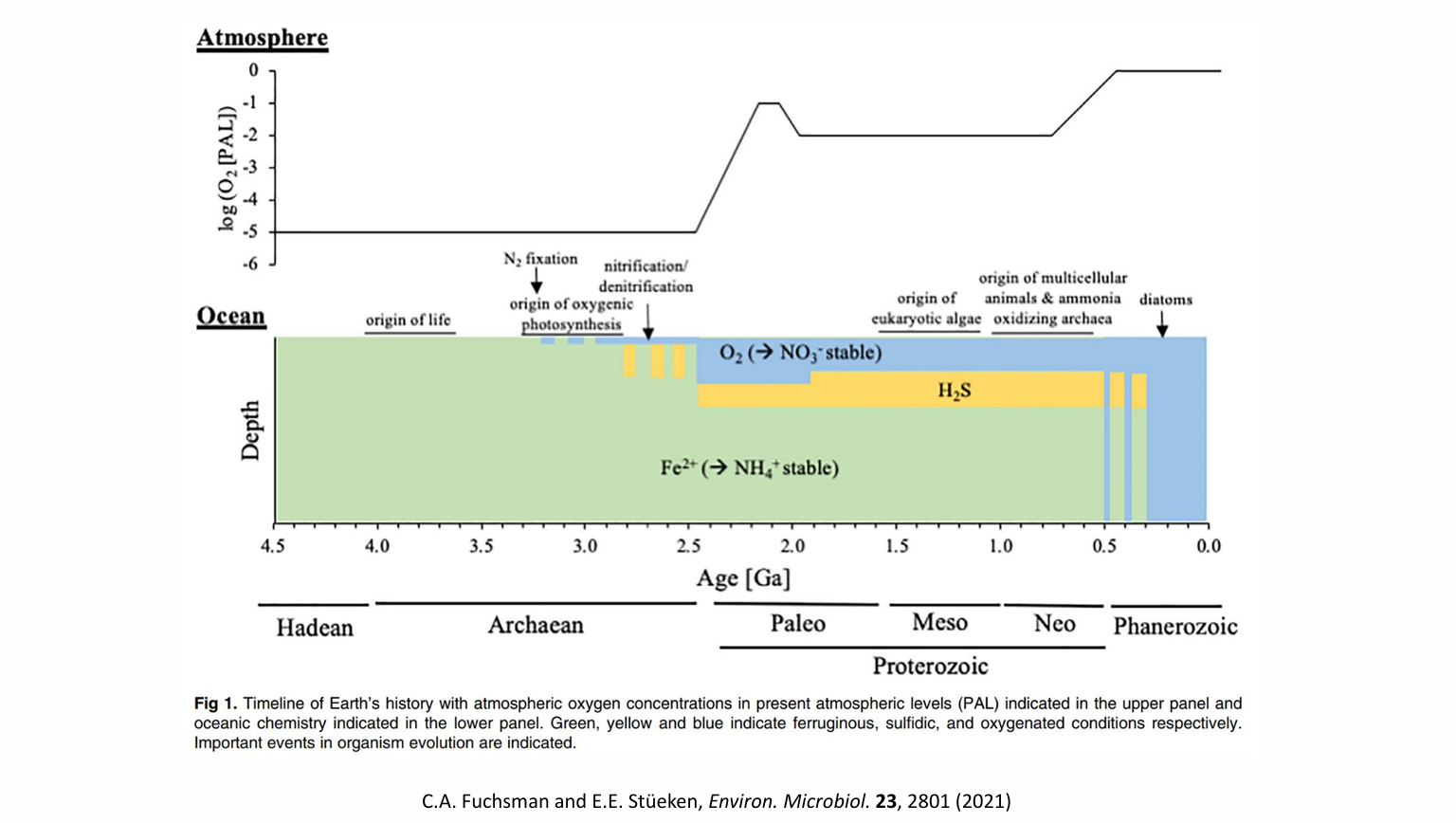
Earth surface has become more and more oxidized over time as evident from the geological record [6-8]. Hydrogen escape may have played an important role in the early stage [9, 10]. Subduction of organic matter and pyrite into the mantle may be the sustained driving force [11, 12], with even deeper mantle-crustal processes playing a role [13]. A consequence of these processes is the rising of O2 concentration (pO2) in the atmosphere, followed by oxygenation of the shallow oceans and eventually the deep oceans. Associated changes include the scavenge of Fe(III) by O2 and Fe(II) by sulfide as well as the increase of SO42- concentration in the ocean overtime. Various positive and negative feedback loops are in play.
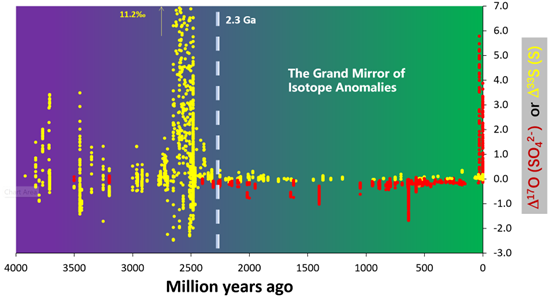
In the midst of this complex and irreversible evolution, there is a threshold of atmospheric O2 concentration and therefore that of total atmospheric ozone column thickness. Below that threshold, deep ultraviolet can reach the ground or the troposphere and non-mass-dependent (NMD) sulfur isotope fractionation can be readily generated by the photolysis of volcanic SO2 at Earth surface and the signatures (i.e., non-zero Δ33S values) preserved in rock record. Above that threshold, NMD sulfur isotope signatures can no longer be generated readily in the troposphere and even more difficult to be preserved in rock record due to increasing intensity of microbial sulfur redox cycling. Instead, NMD oxygen isotope signatures appear in the atmosphere. This is because below the threshold, atmospheric CO2-O2-O3 network reaction is insignificant; above the threshold, atmospheric O2 bears non-mass-dependently 17O-depleted signature while O3 bears 17O-positive signature (i.e., non-zero Δ17O values) [14]. This pO2 threshold is likely at 10-5 Present Atmospheric Level based on atmospheric chemistry models [15, 16]. The transition was coined the Great Oxidation Event (GOE), occurring at ~2.3 Ga [7], when atmospheric CO2-O2-O3 network reaction was becoming significant. Thus, the Δ33S and Δ17O of atmospheric compounds should constitute a mirror image of each other divided at ~2.3 Ga over the now ~4.6 billion-year Earth history, i.e., we will see a Grand Mirror of Isotope Anomalies over the axis of the entire geological time (Fig. 1). The GMIA hypothesis hinges upon the existence of a pO2 threshold and its impacts on the underlying chemistries that produce and preserve the NMD 33S and 17O anomalies.
The GMIA hypothesis has to imprint its atmospheric signatures into minerals and rocks in order to be preserved and tested. Making the atmosphere-rock connection is not straightforward. That’s where the geochemists step in. The proposed mechanism [6] is that when SO2 is photolyzed by UV in the atmosphere, elemental sulfur and S(VI)O3 are produced. The water-insoluble elemental sulfur, S8 mostly, undergoes further disproportionation to generate S- and subsequently to solid FeS/FeS2 and S(IV) species while the S(VI)O3 hydrolyzes to water-soluble SO42- and subsequently locked in minerals like barite or anhydrite. The sulfide minerals bear Δ33S-positive while the SO42- bears Δ33S-negative signatures. Importantly, due to the apparent dearth of sulfide oxidation and sulfate reduction in early Earth environment, these contrasting signatures were not homogenized or erased.
On the oxygen side, stratospheric CO2-O2-O3 reaction results in O3 and CO2 bearing distinct positive Δ17O values while O2 bearing negative Δ17O value [17]. Stratospheric 17O-depleted O2 mixes with photosynthetically produced, 17O-normal O2 to make 17O-depleted tropospheric O2. Tropospheric O3 also bears large positive Δ17O values. When sulfate forms via the oxidation of sulfide minerals or SO2 gas, sulfate oxygen can carry tropospheric O2 or O3 signatures. Sulfate minerals are the most reliable recorder of the past atmospheric O2 and O3 signatures [18].
Although much of the geological time axis remains to be filled, the rock record has revealed a broad pattern of the expected GMIA. With a vertical 33S- or 17O-anomaly axis centered at ~2.3 Ga on the time axis, we can divide the diagram into four quadrants. The 2nd, the positive Δ33S quadrant before 2.3 Ga has been occupied by sedimentary pyrite. We see 33S anomalies in the sedimentary sulfides as old as the oldest rocks on Earth [19]. Only sporadic barite occurrences before 3.2 Ga and pyrite clusters around 2.5 Ga have occupied the 3rd, the Δ33S-negative quadrant before 2.3 Ga. Recent discovery of NMD 17O depletion in sulfate [20-23] has filled most of the time intervals in the 4th, the Δ17O-negative quadrant after 2.3 Ga, while only younger-than-30-Ma volcanic-ash-bed [24] or hyperarid sulfate deposits [25, 26] dotted the 1st, the Δ17O-positive quadrant after 2.3 Ga. The lack of positive Δ17O records in geological history is apparently a preservation issue: Δ17O-positive sulfate minerals can only accumulate in arid and hyperarid continental settings which can hardly survive erosion in a tectonically active planet.
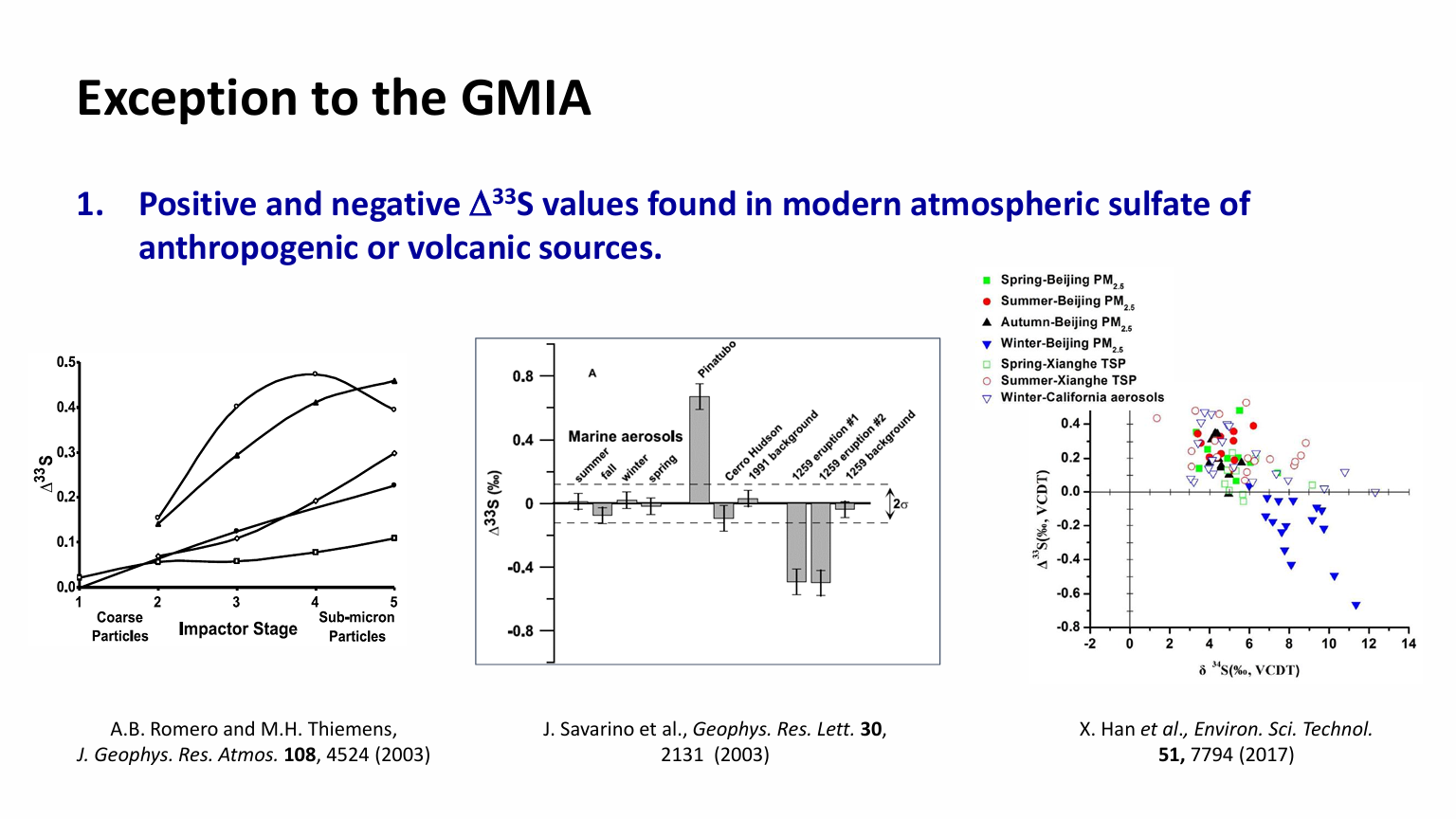
Despite the confirmed pattern of the GMIA, a few exceptions to the GMIA do exist and their resolution may strengthen or potentially weaken the GMIA hypothesis. The most outstanding piece of the exceptions is the positive and negative Δ33S values found in modern atmospheric sulfate of anthropogenic or volcanic sources [27-31]. Sulfur from the late Ordovician black shales in South China also display distinctly positive Δ33S values [32]. Some of the Paleoproterozoic [33] and the latest Permian pyrite grains [34] were reported to be distinctly positive or negative in Δ33S values. The modern atmospheric 33S anomalies may be explained by the unprecedent high-spatial-temporal sampling resolution that has captured the reaction-transport effects which are otherwise homogenized in geological samples. The discoveries of Proterozoic and Phanerozoic sedimentary 33S anomalies, while puzzling, have not yet constituted a critical challenge to the existing pO2 threshold paradigm. The GMIA hypothesis will be continuously tested and enhanced with new data.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (42494851, 42494852).
References
[1] R.N. Mitchell, N. Zhang, J. Salminen, Y. Liu, C.J. Spencer, B. Steinberger, J.B. Murphy, and Z.-X. Li, The supercontinent cycle, Nat. Rev. Earth Environ. 2, 358 (2021).
[2] R.D. Nance and J.B. Murphy, Origins of the supercontinent cycle, Geosci. Front. 4, 439 (2013).
[3] M. Domeier, V. Magni, M.W. Hounslow, and T.H. Torsvik, Episodic zircon age spectra mimic fluctuations in subduction, Sci. Rep. 8, 17471 (2018).
[4] M.R. Rampino, K. Caldeira, and S. Rodriguez, Sixteen mass extinctions of the past 541 My correlated with 15 pulses of Large Igneous Province (LIP) volcanism and the 4 largest extraterrestrial impacts, Glob. Planet. Change 234, 104369 (2024).
[5] S.E. Grasby and D.P.G. Bond, How large igneous provinces have killed most life on Earth—numerous times, Elements 19, 276 (2023).
[6] J. Farquhar, H. Bao, and M. Thiemens, Atmospheric influence of Earth's earliest sulfur cycle, Science 289, 756 (2000).
[7] H.D. Holland, The oxygenation of the atmosphere and oceans, Philos. Trans. R. Soc. B 361, 903 (2006).
[8] C.A. Fuchsman and E.E. Stüeken, Using modern low-oxygen marine ecosystems to understand the nitrogen cycle of the Paleo- and Mesoproterozoic oceans, Environ. Microbiol. 23, 2801 (2021).
[9] D.C. Catling and M.W. Claire, How Earth's atmosphere evolved to an oxic state: a status report, Earth Planet. Sci. Lett. 237, 1 (2005).
[10] K.J. Zahnle, D.C. Catling, and M.W. Claire, The rise of oxygen and the hydrogen hourglass, Chem. Geol. 362, 26 (2013).
[11] M.S. Duncan and R. Dasgupta, Rise of Earth’s atmospheric oxygen controlled by efficient subduction of organic carbon, Nat. Geosci. 10, 387 (2017).
[12] D.A. Stolper, J.A. Higgins, and L.A. Derry, The role of the solid earth in regulating atmospheric O₂ levels, Am. J. Sci. 321, 1381 (2021).
[13] M.M. Hirschmann, The deep Earth oxygen cycle: mass balance considerations on the origin and evolution of mantle and surface oxidative reservoirs, Earth Planet. Sci. Lett. 619, 118311 (2023).
[14] Y.L. Yung, A.Y.T. Lee, F.W. Irion, W.B. DeMore, and J. Wen, Carbon dioxide in the atmosphere: isotopic exchange with ozone and its use as a tracer in the middle atmosphere, J. Geophys. Res. Atmos. 102, 10857 (1997).
[15] N.F. Wogan, D.C. Catling, K.J. Zahnle, and M.W. Claire, Rapid timescale for an oxic transition during the Great Oxidation Event and the instability of low atmospheric O₂, Proc. Natl. Acad. Sci. U.S.A. 119, e2205618119 (2022).
[16] A.A. Pavlov and J.F. Kasting, Mass-independent fractionation of sulfur isotopes in Archean sediments: strong evidence for an anoxic Archean atmosphere, Astrobiology 2, 27 (2002).
[17] B. Luz, E. Barkan, M.L. Bender, M.H. Thiemens, and K.A. Boering, Triple-isotope composition of atmospheric oxygen as a tracer of biosphere productivity, Nature 400, 547 (1999).
[18] H. Bao, Sulfate: a time capsule for Earth’s O₂, O₃, and H₂O, Chem. Geol. 395, 108 (2015).
[19] E. Thomassot, J. O'Neil, D. Francis, P. Cartigny, and B.A. Wing, Atmospheric record in the Hadean Eon from multiple sulfur isotope measurements in Nuvvuagittuq Greenstone Belt (Nunavik, Quebec), Proc. Natl. Acad. Sci. U.S.A. 112, 707 (2015).
[20] H. Wang, C. Li, Y. Peng, J. Zhang, M. Cheng, X. Cao, W. Qie, Z. Zhang, M.S. Dodd, M. Hou, M. Wallace, A.V.S. Hood, T.W. Lyons, and H. Bao, Two-billion-year transitional oxygenation of the Earth’s surface, Nature 645, 665 (2025).
[21] H. Wang, Y. Peng, C. Li, X. Cao, M. Cheng, and H. Bao, Sulfate triple-oxygen-isotope evidence confirming oceanic oxygenation 570 million years ago, Nat. Commun. 14, 4315 (2023).
[22] H. Bao, J.R. Lyons, and C. Zhou, Triple oxygen isotope evidence for elevated CO₂ levels after a Neoproterozoic glaciation, Nature 453, 504 (2008).
[23] P.W. Crockford, J.A. Hayles, H. Bao, N.J. Planavsky, A. Bekker, P.W. Fralick, G.P. Halverson, T.H. Bui, Y. Peng, and B.A. Wing, Triple oxygen isotope evidence for limited mid-Proterozoic primary productivity, Nature 559, 613 (2018).
[24] H. Bao, S. Yu, and D.Q. Tong, Massive volcanic SO₂ oxidation and sulphate aerosol deposition in Cenozoic North America, Nature 465, 909 (2010).
[25] H. Bao and D.R. Marchant, Quantifying sulfate components and their variations in soils of the McMurdo Dry Valleys, Antarctica, J. Geophys. Res. 111, D16301 (2006).
[26] T. Sun, H. Bao, M. Reich, and S.R. Hemming, More than ten million years of hyper-aridity recorded in the Atacama Gravels, Geochim. Cosmochim. Acta 227, 123 (2018).
[27] A.B. Romero and M.H. Thiemens, Mass-independent sulfur isotopic compositions in present-day sulfate aerosols, J. Geophys. Res. Atmos. 108, 4524 (2003).
[28] X. Han, Q. Guo, H. Strauss, C. Liu, J. Hu, Z. Guo, R. Wei, M. Peters, L. Tian, and J. Kong, Multiple sulfur isotope constraints on sources and formation processes of sulfate in Beijing PM₂.₅ aerosol, Environ. Sci. Technol. 51, 7794 (2017).
[29] I. Genot, D. Au Yang, E. Martin, P. Cartigny, E. Legendre, and M. De Rafelis, Oxygen and sulfur mass-independent isotopic signatures in black crusts: the complementary negative Δ³³S reservoir of sulfate aerosols?, Atmos. Chem. Phys. 20, 4255 (2020).
[30] J. Savarino, A. Romero, J. Cole-Dai, S. Bekki, and M.H. Thiemens, UV-induced mass-independent sulfur isotope fractionation in stratospheric volcanic sulfate, Geophys. Res. Lett. 30, 2131 (2003).
[31] M. Baroni, M.H. Thiemens, R.J. Delmas, and J. Savarino, Mass-independent sulfur isotopic compositions in stratospheric volcanic eruptions, Science 315, 84 (2007).
[32] D. Hu, M. Li, X. Zhang, A.V. Turchyn, Y. Gong, and Y. Shen, Large mass-independent sulfur isotope anomalies link stratospheric volcanism to the Late Ordovician mass extinction, Nat. Commun. 11, 2297 (2020).
[33] S.A. Young, K. Loukola-Ruskeeniemi, and L.M. Pratt, Reactions of hydrothermal solutions with organic matter in Paleoproterozoic black shales at Talvivaara, Finland: evidence from multiple sulfur isotopes, Earth Planet. Sci. Lett. 367, 1 (2013).
[34] R. Li, S.-Z. Shen, X.-P. Xia, B. Xiao, Y. Feng, and H. Chen, Atmospheric ozone destruction and the end-Permian crisis: evidence from multiple sulfur isotopes, Chem. Geol. 647, 121936 (2024).



